Abstract
Background: Despite advances in the therapeutics of Sickle Cell Disease (SCD), vaso-occlusion, painful crisis, and other complications (such as delayed hemolytic transfusion reaction/DHTR) remain a major cause of morbidity and mortality. Recent research data suggest complement activation, especially during SCD complications. These advances led to recommendation of complement inhibition in patients with DHTR and ongoing hyperhemolysis. However, limited data exist about other SCD complications.
Hypothesis: We hypothesized that novel functional assays will be useful in detecting complement activation in SCD, especially during complications, and that these patients present distinct clinical or genetic features that have not been investigated yet.
Methods: We enrolled consecutive SCD patients from four Thalassemia Units, meeting the eligibility criteria: (i) age > 16 years, (ii) established SCD diagnosis and (iii) willingness to participate in the study and sign a written informed consent. We collected samples at enrolment and at any updates of disease status during 1-year follow-up (initiation of hydroxyurea, vaso-occlusive crisis, hemolytic reactions, nephropathy, or TMA) or at last follow-up if no update was observed. Complement activation was detected using soluble C5b-9 (Quidel kit), and the modified Ham test. Values above 245 ng/ml have been established in our laboratory as above normal limits for C5b-9, and 20% for modified Ham test, respectively. The latter is a cell proliferation assay based on the susceptibility of a PNH (paroxysmal nocturnal hemoglobinuria)-like cell line to complement-activated serum. We also utilized next generation sequencing (NGS) analysis with a customized panel (complement factor H/CFH, CFH-related, CFI, CFB, CFD, C3, CD55, C5, CD46, thombomodulin/THBD, ADAMTS13, MiniSeq, Illumina). Identified variants were evaluated by using 4 bioinformatic tools. Variants predicted to be deleterious from at least 2 tools (50%), were assumed as pathogenic and were compared with the 2018 database of complement gene variants.
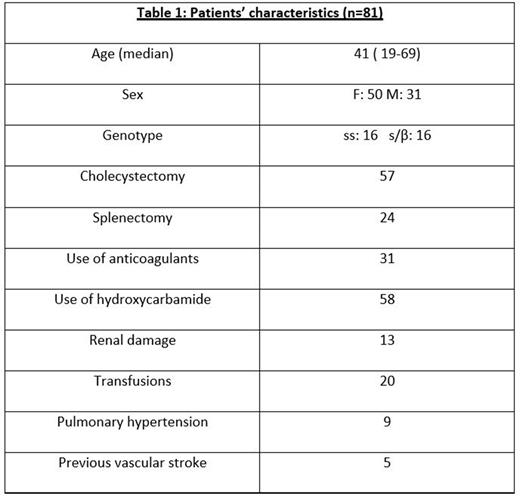
Results: We studied 81 adult SCD patients (Table 1). Among them, 23 presented a disease update during follow-up (17 vaso-occlusive crises and 6 proteinuria). Patients with SCD complications during follow-up had excessive complement activation detected even from steady state with increased C5b-9 (7/23, 30%, p=0.028) and positive modified Ham test (5/23, 21%, p=0.001). During follow-up, C5b-9 significantly increased (p<0.001), particularly in patients with complications (p=0.046). Modified Ham test positivity remained the same.
Regarding genetics, we detected 25 rare variants (Minor Allele Frequency < 1%). Only 3 of them found in 3 patients were also characterized as pathogenic: rs117793540 in C3, rs1800579 in THBD, and rs143568784 in ADAMTS13. We also documented 5 additional deleterious variants in 12 patients: rs144082872 in CFI, rs12614 in CFB, rs28503257, rs145825553 and rs28647808 in ADAMTS13. Patients were heterozygous, except for 1 homozygous for rs28647808. Patients with deleterious variants had increased complement activation by functional assays, except for 2 patients with CFB and 2 with ADAMTS13 variants. Interestingly, patients with a combination of a deleterious variant had a significantly higher transfusion dependency rate (65% versus 35%, p=0.031).
Conclusion We have shown for the first time prospectively that complement is activated even from steady state in patients that develop SCD complication, with significant increase during complications. Complement-related genetic variants undeline this predisposition to complement activation in certain patients. Considering safety and efficacy of complement inhibitors in other complementopathies and preliminary data from SCD, our study highlights useful tools to early detect patients that might benefit from complement inhibition.
Disclosures
Diamantidis:Bristol Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Honoraria; GENESIS Pharma SA: Honoraria, Research Funding; Uni-Pharma: Honoraria; Ionis Pharmaceuticals: Other: Clinical Trial Research Support (PI); NOVARTIS: Other: Clinical Trial Research Support (PI); Vifor International: Other: Clinical Trial Research Support (PI); Forma Therapeutics Inc. and Synteract: Other: Clinical Trial Research Support (PI). Brodsky:Alexion: Honoraria. Gavriilaki:Gilead: Honoraria; Jazz: Research Funding; Sanofi: Honoraria; Omeros: Honoraria; Alexion: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal